  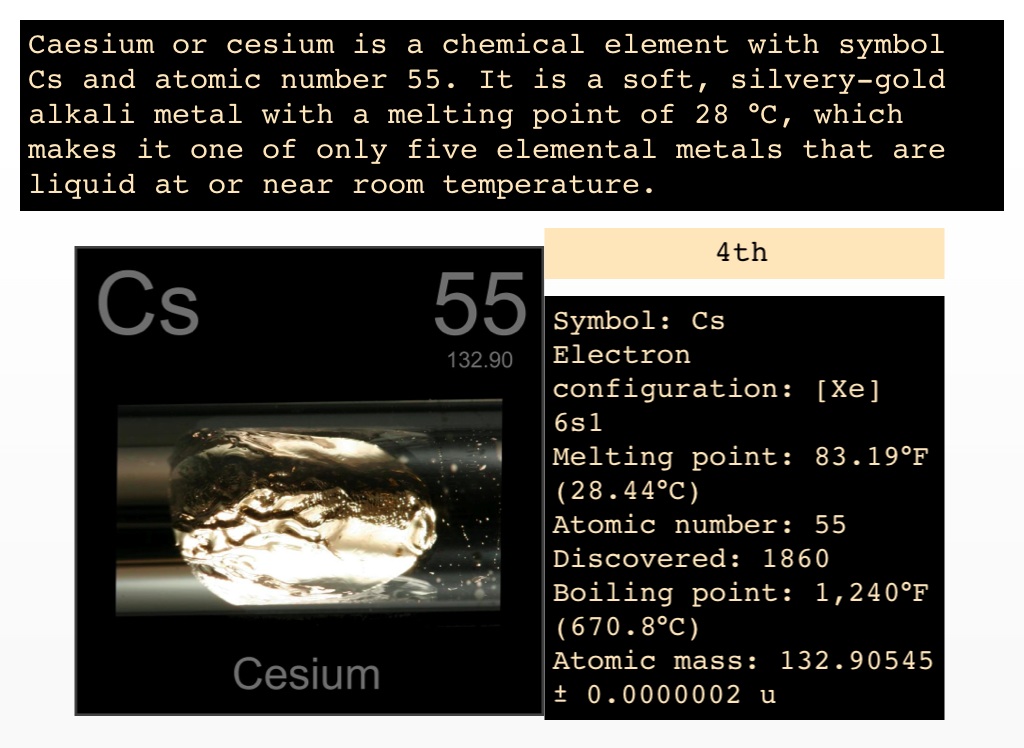
Caesium is the second most electropositive and alkaline of the chemical elements and has the second lowest ionization potential (after francium). This metal is silvery gold in color and is both soft and ductile. The emission spectrum of caesium has two bright lines in the blue part of the spectrum along with several other lines in the red, yellow, and green. The variant spelling cesium is used especially in North American English, and the IUPAC has recognized it as a variant spelling since 1993, but caesium is the spelling used by the IUPAC. This element is most notably used in atomic clocks. It is a soft silvery-gold alkali metal with a melting point of 28 ☌ (83 ☏), which makes it one of the metals that are liquid at or near room temperature, along with rubidium ( 39 ☌ ), francium ( 27 ☌ ), mercury ( −39 ☌ ), and gallium ( 30 ☌ ). Template:Elementbox isotopes stable Template:Elementbox isotopes decay2 Template:Elementbox isotopes decay Template:Elementbox isotopes decay Template:Elementbox isotopes end Template:Elementbox footerĬaesium or cesium ( Template:PronEng) is the chemical element with the symbol Cs and atomic number 55. ! colspan="2" style="background:#ff6666 color:black" | Selected isotopes Density: 1.Template:Elementbox header Template:Elementbox series Template:Elementbox groupperiodblock Template:Elementbox appearance img Template:Elementbox atomicmass gpm Template:Elementbox econfig Template:Elementbox epershell Template:Elementbox section physicalprop Template:Elementbox phase Template:Elementbox density gpcm3nrt Template:Elementbox densityliq gpcm3mp Template:Elementbox meltingpoint Template:Elementbox boilingpoint Template:Elementbox criticalpoint Template:Elementbox heatfusion kjpmol Template:Elementbox heatvaporiz kjpmol Template:Elementbox heatcapacity jpmolkat25 Template:Elementbox vaporpressure katpa Template:Elementbox section atomicprop Template:Elementbox crystalstruct Template:Elementbox oxistates Template:Elementbox electroneg pauling Template:Elementbox ionizationenergies3 Template:Elementbox atomicradius pm Template:Elementbox atomicradiuscalc pm Template:Elementbox covalentradius pm Template:Elementbox section miscellaneous Template:Elementbox magnetic Template:Elementbox eresist ohmmat20 Template:Elementbox thermalcond wpmkat300k Template:Elementbox thermalexpansion umpmkat25 Template:Elementbox youngsmodulus gpa Template:Elementbox bulkmodulus gpa Template:Elementbox mohshardness Template:Elementbox brinellhardness mpa Template:Elementbox cas number.Here are a few important facts about Cesium. Since cesium has photoemissive properties, converting light to electron flow, it has use in photoelectric cells to convert sunlight into electricity. A cesium-based atomic clock will lose one-second per 100 million years. Since cesium combines readily with oxygen, it has uses as a getter material, which combines with and removes trace gases from vacuum tubes.Ĭesium also has some uses in extremely accurate atomic clocks. Its largest use in modern times is in cesium formate drilling fluids utilized in oil well drilling.  
Due to cesium having the second-lowest melting point of all metallic elements, its uses are quite limited. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
